Atomic Orbital Diagram For Chlorine
Draw the atomic orbital diagram for chlorine. Construct the orbital diagram of each atom or ion.
The p orbital can hold up to six electrons.
Atomic orbital diagram for chlorine. Chlorine cl has an atomic mass of 17. The final ring or shell of electrons contains the typical number of valence electrons for an atom of that element. In a chlorine atom which subshells contain valence electrons.
The green dot represents the location of a chlorine nucleus significantly enlarged so that you can see it. Since the 3s if now full well move to the 3p where well place the remaining five electrons. If the two 1s orbitals are not in phase a node between them causes a jump in energy the σ orbital.
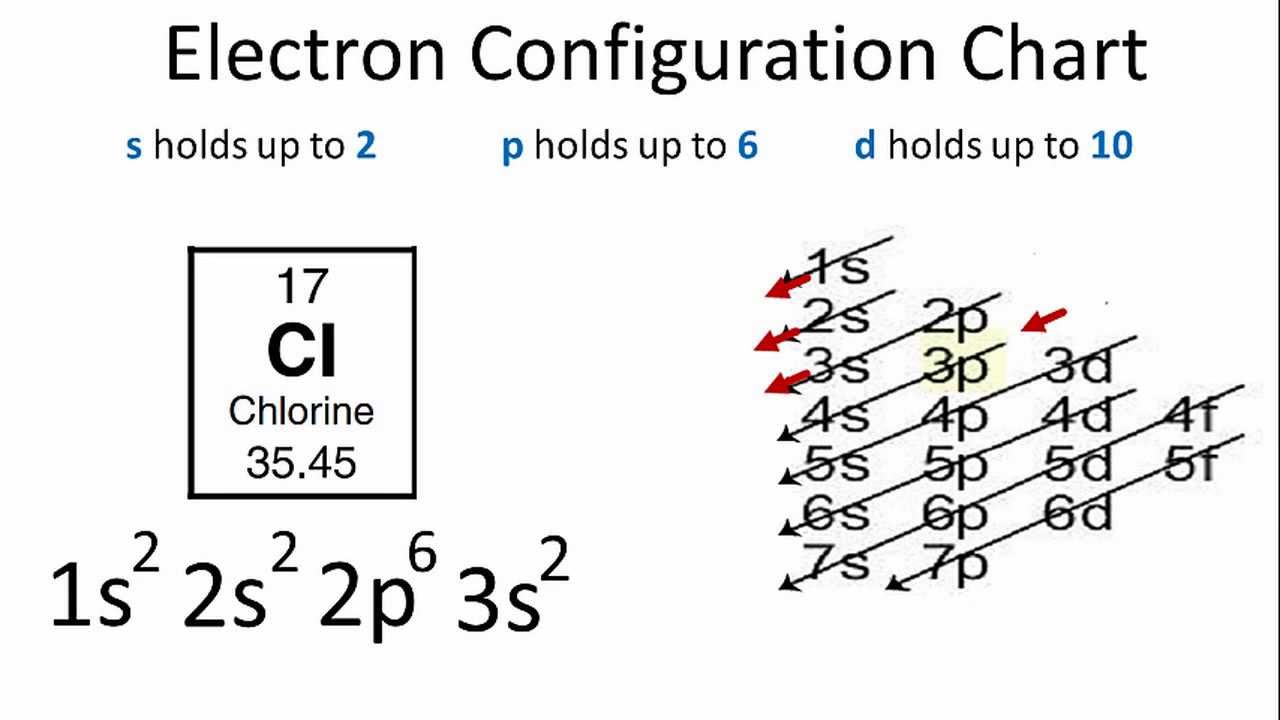
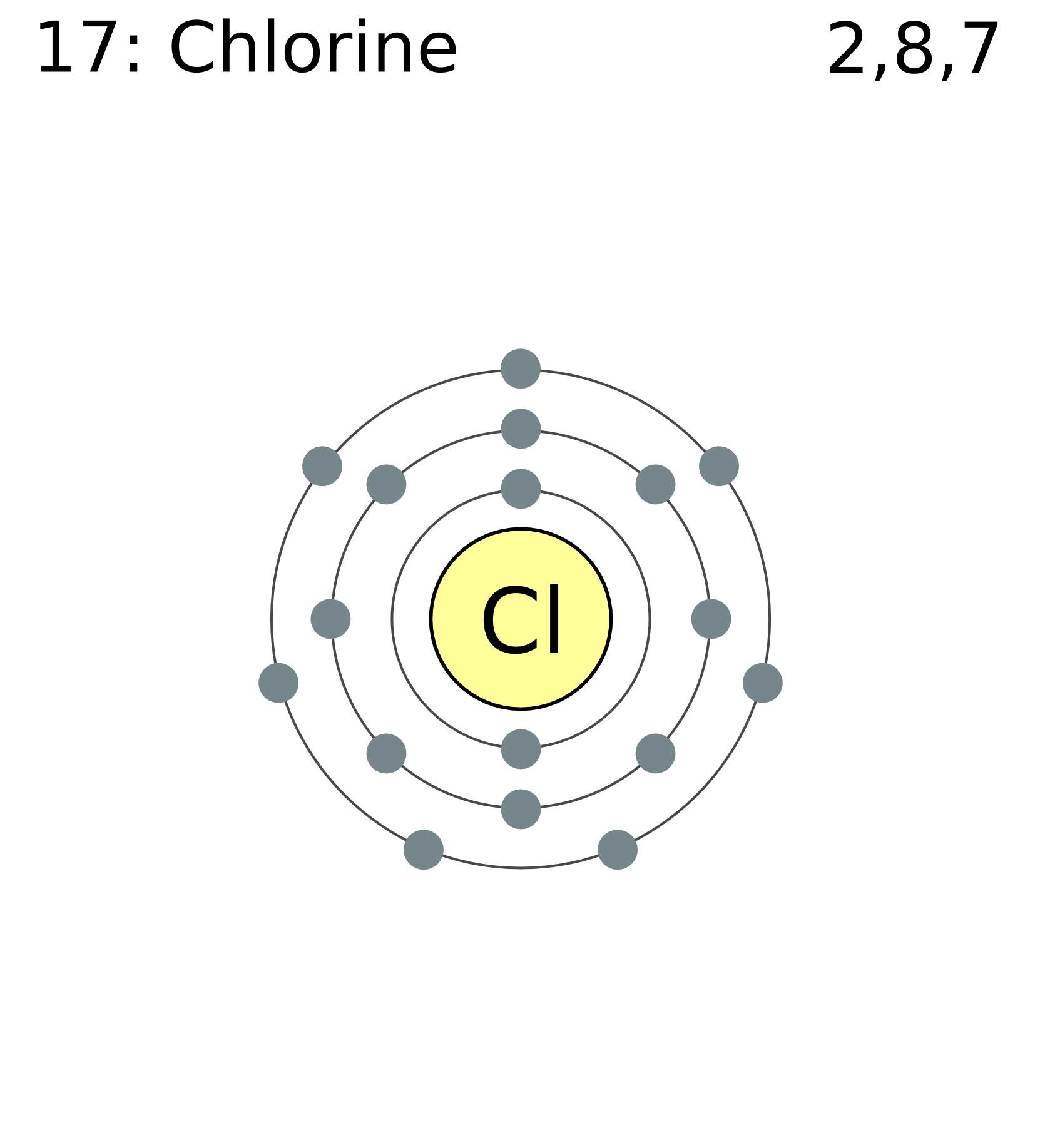
Therefore the chlorine electron configuration will be 1s22s22p63s23p5. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atoms nucleus. The electron shells are shown moving outward from the nucleus.
Create the atomic orbital diagram for chlorine. Show transcribed image text create the atomic orbital diagram for chlorine. Electron configurations and orbital diagrams key draw orbital diagrams for the following elements.
Find out about its chemical and physical properties states energy electrons oxidation and more. Two atomic orbitals in phase create a larger electron density which leads to the σ orbital. Draw the atomic orbital diagram for chlorine.
In atomic theory and quantum mechanics an atomic orbital is a mathematical function that describes the wave like behavior of either one electron or a pair of electrons in an atom. How many electrons does a fe atom have in its 3. For each atom diagram the element symbol is listed in the nucleus.
Well put six in the 2p orbital and then put the next two electrons in the 3s. On the right hand side are four pull down menus from which you can choose an orbital to display. The electronic configuration of a ground state chlorine is ne3s23p5 1s22s22p63s23p5.
An orbital diagram is a sketch which shows electron population in atomic orbitals with the electrons spin indicated by up and down arrows. Elements are show from atomic number 1 hydrogen up to 94 plutonium. The superposition of the two 1s atomic orbitals leads to the formation of the σ and σ molecular orbitals.
Create the atomic orbital diagram for nitrogen. Phosphorus 1s 2s 2p 3s 3p 4s 3d 4p 2.
 Chlorine Electron Configuration Youtube
Chlorine Electron Configuration Youtube
Vsepr Clf3 Chlorine Trifluoride
 High School Chemistry Lewis Electron Dot Diagrams Wikibooks Open
High School Chemistry Lewis Electron Dot Diagrams Wikibooks Open
Hf Molecular Orbital Diagram Luxury Molecular Orbital Diagram For
 Combined Dos Of S And P Orbitals Of A B Oxygen Atoms And C
Combined Dos Of S And P Orbitals Of A B Oxygen Atoms And C
 Ground State Electron Configuration Definition Example Video
Ground State Electron Configuration Definition Example Video
Sparknotes Organic Chemistry Atomic Structure Atoms And Atomic
 Orbital Diagram For Chlorine Pics Download
Orbital Diagram For Chlorine Pics Download
Chlorine Orbital Diagram Unique Energy Levels And Electron
 Ground State Orbital Diagram Magnesium Wiring Diagram Schematics
Ground State Orbital Diagram Magnesium Wiring Diagram Schematics
 Orbital Diagram Cl Best Wiring Library
Orbital Diagram Cl Best Wiring Library
 Orbital Diagram Cl Best Wiring Library
Orbital Diagram Cl Best Wiring Library
 7 3 Lewis Symbols And Structures Chemistry
7 3 Lewis Symbols And Structures Chemistry
 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
 8 4 Molecular Orbital Theory Chemistry
8 4 Molecular Orbital Theory Chemistry
 Section 11 4 Electron Configurations And Atomic Properties 1 To
Section 11 4 Electron Configurations And Atomic Properties 1 To
 What Is The Bond Order Of Oxygen Quora
What Is The Bond Order Of Oxygen Quora
 Chlorine Orbital Diagram Theoretical Chemistry Energetic Placement
Chlorine Orbital Diagram Theoretical Chemistry Energetic Placement
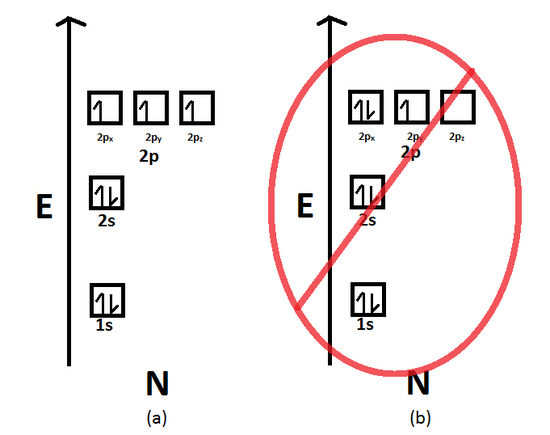 Electron Configuration Chemistry Libretexts
Electron Configuration Chemistry Libretexts
Chlorine Orbital Diagram Chemistry Archive September 08 2016 Air

0 Response to "Atomic Orbital Diagram For Chlorine"
Post a Comment