What Is The Activation Energy For The Reaction In This Energy Diagram
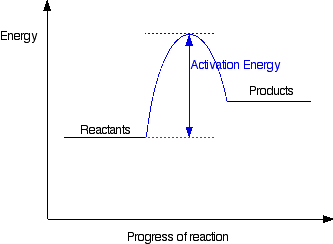
In this diagram the activation energy is signified by the hump in the reaction pathway and is labeled. The activation energy ea of a reaction is measured in joules.
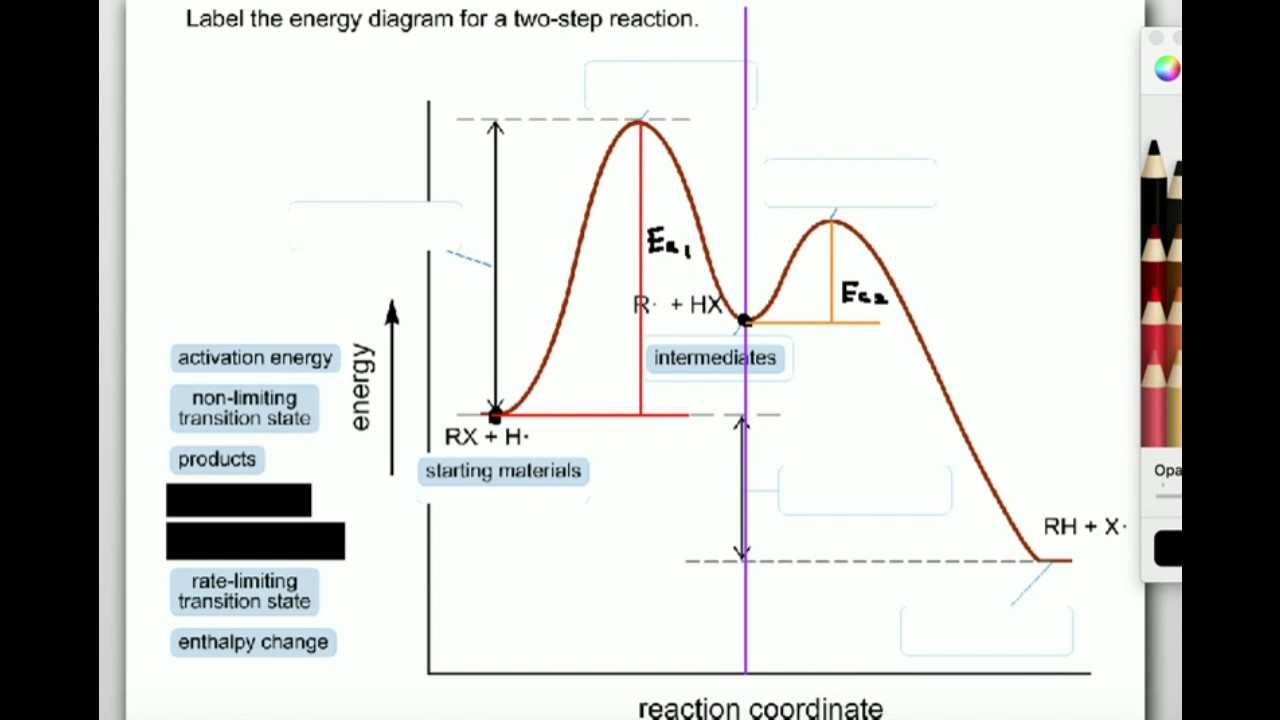 Labeling Parts Of A Reaction Coordinate Diagram Youtube
Labeling Parts Of A Reaction Coordinate Diagram Youtube
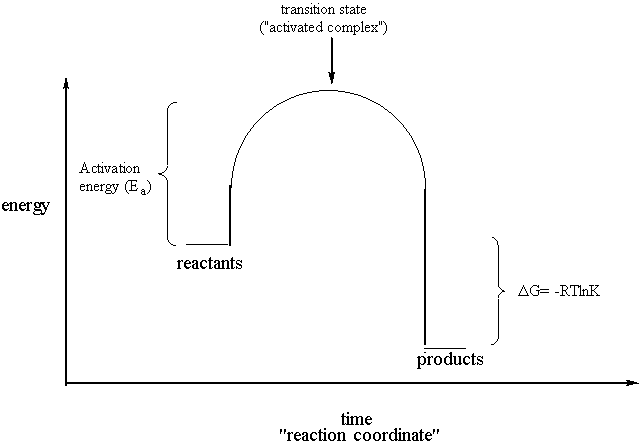
At the peak of the activation energy hump the reactants are in the transition state halfway between being reactants and forming products.
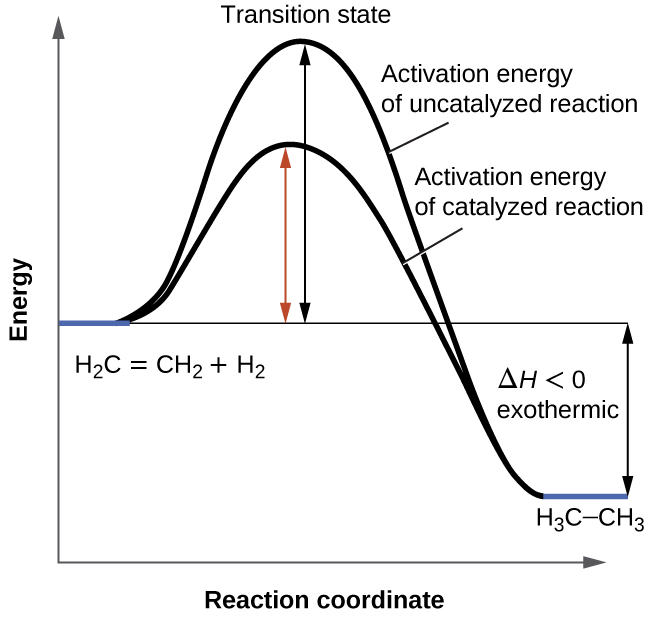
What is the activation energy for the reaction in this energy diagram. The activation energy for the following reaction is 125 kjmol. In energy profile diagram it is the difference in energy from the reactants to topmost peak of the graph. At the peak of the activation energy hump the reactants are in the transition state halfway between being reactants and forming products.
Activation energy is the difference between the starting amount of energy of the reactants and the energy of the activated complex. The activation energy shown in the diagram below is for the forward reaction reactants products which is exergonic. The answer is 125 kjmol 216 kjmol 341 kjmol.
Activation energy of a reaction is the energy required by which the molecules must collide to give a successful product. If the reaction were to proceed in the reverse direction endergonic the transition state would remain the same but the activation energy would be larger. This site might help you.
The activation energy of the reverse reaction should be equal to the activation energy of the forward reaction minus the drive in the reaction delta e. This state is also known as an activated complex. The energy of the reactants of the reverse reaction is 13 kj the activated complex is still at 83 kj.
This state is also known as an activated complex. Select all that apply a catalyst lowers the activation energy. The vertical axis in this diagram represents the free energy of a pair of molecules as a chlorine atom is transferred from one to the other.
A chemical reaction nuclear reaction or various other physical phenomena. In chemistry and physics activation energy is the energy which must be available to a chemical or nuclear system with potential reactants to result in. The horizontal axis represents the the sequence of infinitesimally small changes that must occur to convert the reactants into the products of this reaction.
In this diagram the activation energy is signified by the hump in the reaction pathway and is labeled.
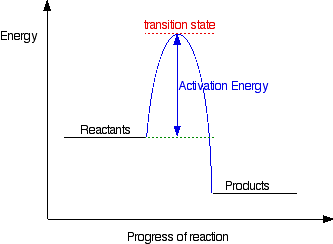 A Look At Energy Profiles For Reactions Chemistry Libretexts
A Look At Energy Profiles For Reactions Chemistry Libretexts
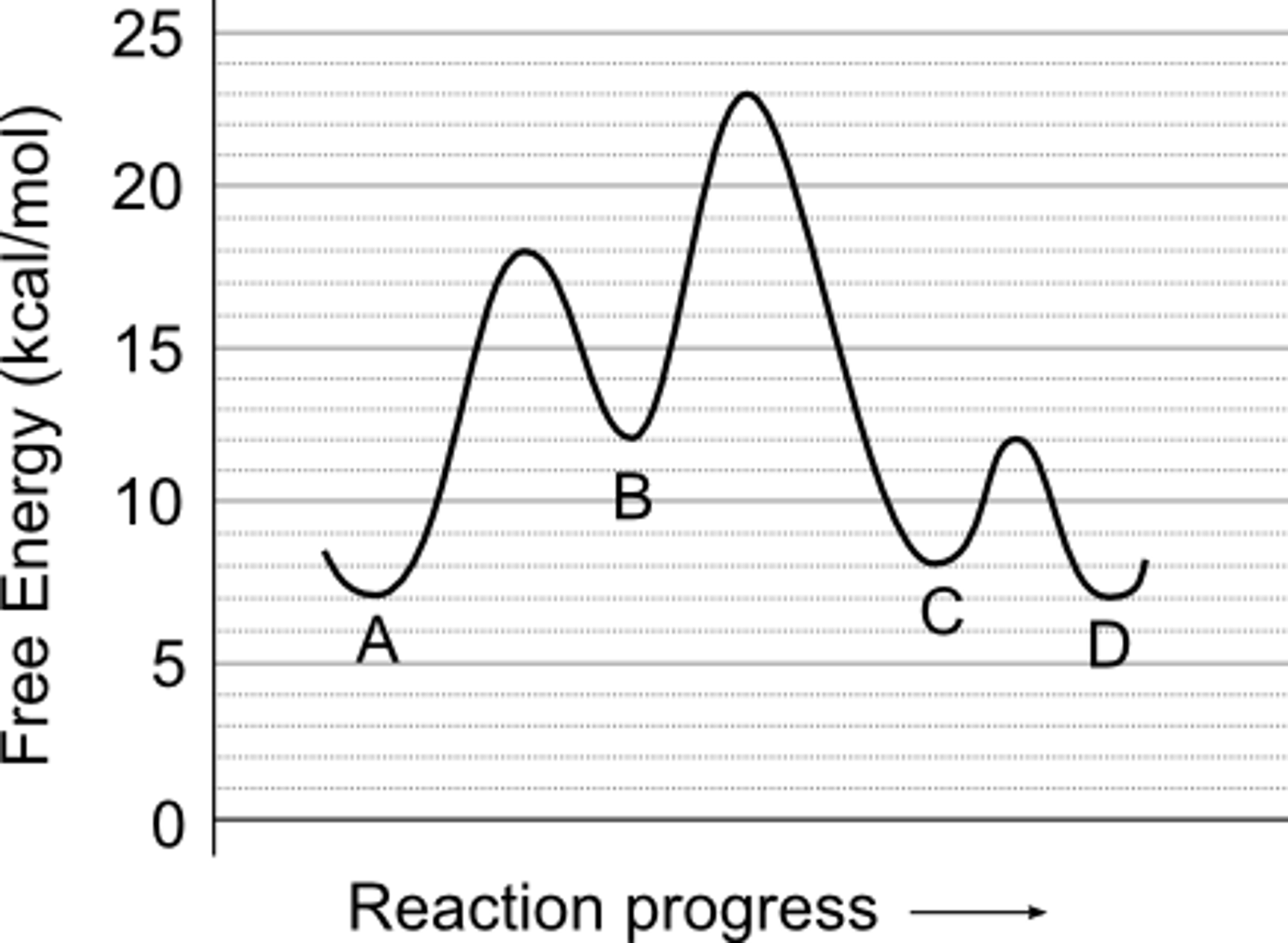 Solved Use The Reaction Energy Diagram Above To Answer Th
Solved Use The Reaction Energy Diagram Above To Answer Th
 Activation Energy Gcse Wiki Fandom Powered By Wikia
Activation Energy Gcse Wiki Fandom Powered By Wikia
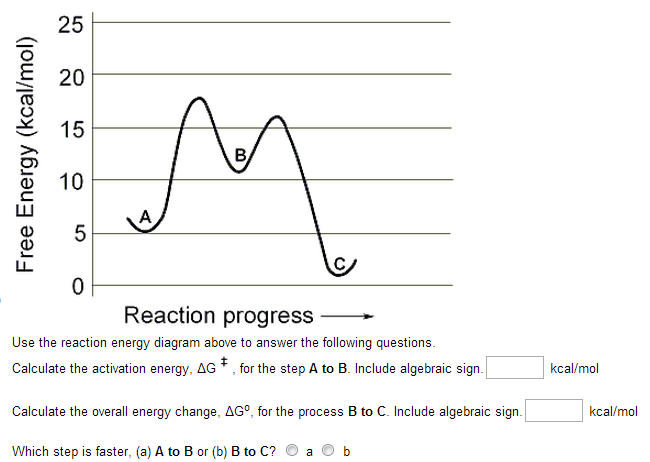 Solved Use The Reaction Energy Diagram Above To Answer Th
Solved Use The Reaction Energy Diagram Above To Answer Th
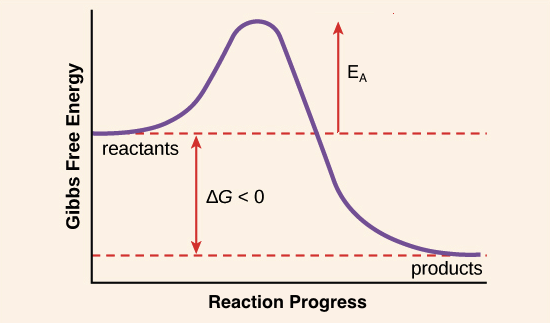 Activation Energy Article Enzymes Khan Academy
Activation Energy Article Enzymes Khan Academy
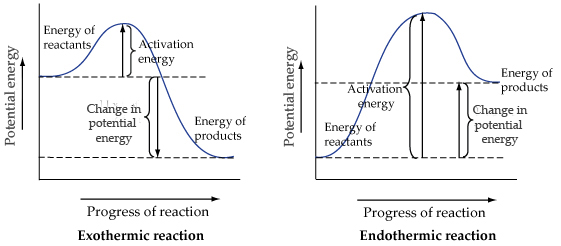 Solved The Reaction Energy Diagrams For An Endothermic An
Solved The Reaction Energy Diagrams For An Endothermic An
What Is The Activation Energy For A Reverse Reaction Quora
Transition States And Activation Energy Open Textbooks For Hong Kong
 Chem 1180 13 5 13 6 Potential Energy Diagrams Arrhenius Equation
Chem 1180 13 5 13 6 Potential Energy Diagrams Arrhenius Equation
 How Can I Represent The Activation Energy In A Potential Energy
How Can I Represent The Activation Energy In A Potential Energy
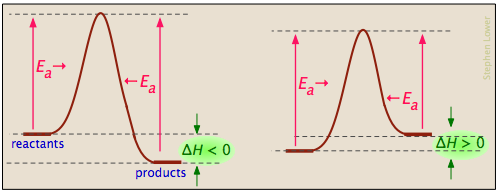 Basics Of Reaction Profiles Chemistry Libretexts
Basics Of Reaction Profiles Chemistry Libretexts
Gcse Chemistry What Are Energy Level Diagrams What Is The
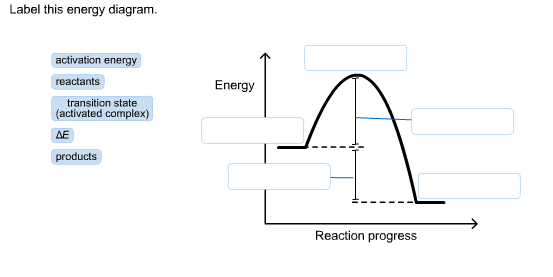 Solved Label This Energy Diagram Activation Energy React
Solved Label This Energy Diagram Activation Energy React
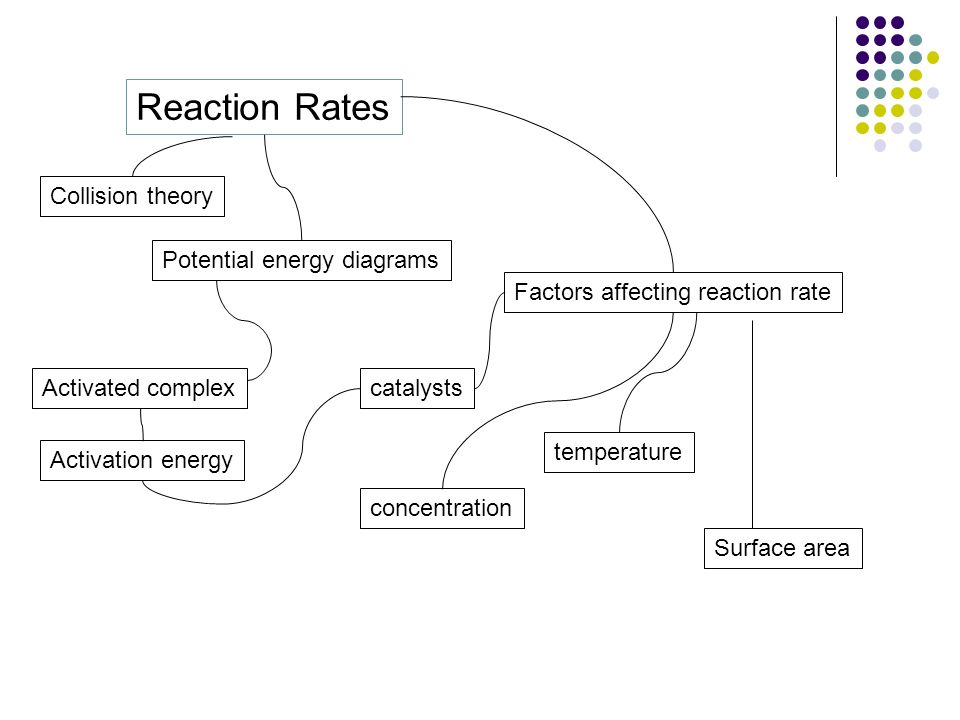 Kinetics Reaction Rates Collision Theory Factors Affecting Reaction
Kinetics Reaction Rates Collision Theory Factors Affecting Reaction
 Potential Energy Diagram With Without Catalyst In A Hypothetical
Potential Energy Diagram With Without Catalyst In A Hypothetical
0 Response to "What Is The Activation Energy For The Reaction In This Energy Diagram"
Post a Comment