Does This Diagram Represent An Increase Or Decrease In The Internal Energy Of The System
Chapter 24 thermodynamics review. If work is done on a system does the internal energy of the system increase or decrease.
The First Law Of Thermodynamics Closed Systems Heat Transfer
A does this diagram represent an increase or decrease in the internal energy of the system.
Does this diagram represent an increase or decrease in the internal energy of the system. Thermodynamics reading check and hw. If work is done by a system does the internal energy of the system increase or decrease. The change in internal energy δetexttip delta e delta e is positive if the system absorbs energy and it is negative if the system releases energy.
A does this diagram represent an increase or decrease in the internal energy of the system. B q 150 kj and w 657 j. Incorrectly answering a question in a part or hint.
The first law of thermodynamics energy cannot be created nor destroyed but it can be transferred between a system and its surroundings. B what sign is given to e for this process. Originproblemid1097921descriptionconsider the energy diagram shown in the from chemistry 151 at rio salado community college.
If work is done adiabatically on a system will the internal energy of the system increase or decrease. If work is done by a system will the internal energy of the system increase or decrease. Calculate delta e energy and determine whether the process is endothermic or exothermic for the follwing cases.
Increase decrease what sign i. C the system releases 575 kj of heat while doing 225 kj of work on the surroundings. B what sign is given to e for this process.
53 consider the accompanying energy diagram. They will both decrease. C if there is no work associated with the process is it exothermic or endothermic.
C if there is no work associated with the process is it exothermic or endothermic. A a system absorbs 105 kj of heat from its surroundings while doing 29 kj of work on the surroundings. Consider the accompanying energy diagram.
Bike pump use warms up. Answer to does this diagram represent an increase or decrease in the internal energy of the system. Diagram represent an increase of internal energy of the system e eproduct ereactant eproduct is bigger than ereactant so e is positive if no work is associated with the process the reaction is.
 Solved Consider The Energy Diagram Shown In The Figure F
Solved Consider The Energy Diagram Shown In The Figure F
The First Law Of Thermodynamics

Helmholtz And Gibbs Free Energies
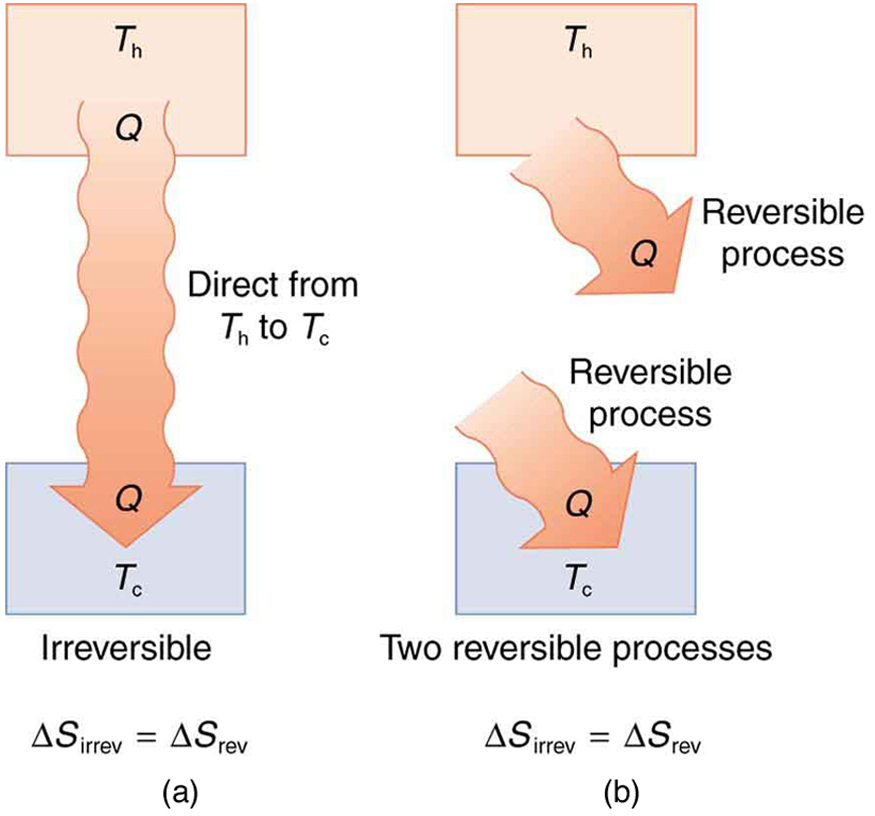 Entropy And The Second Law Of Thermodynamics Disorder And The
Entropy And The Second Law Of Thermodynamics Disorder And The
Chemistry The Central Science Chapter 5 Section 2
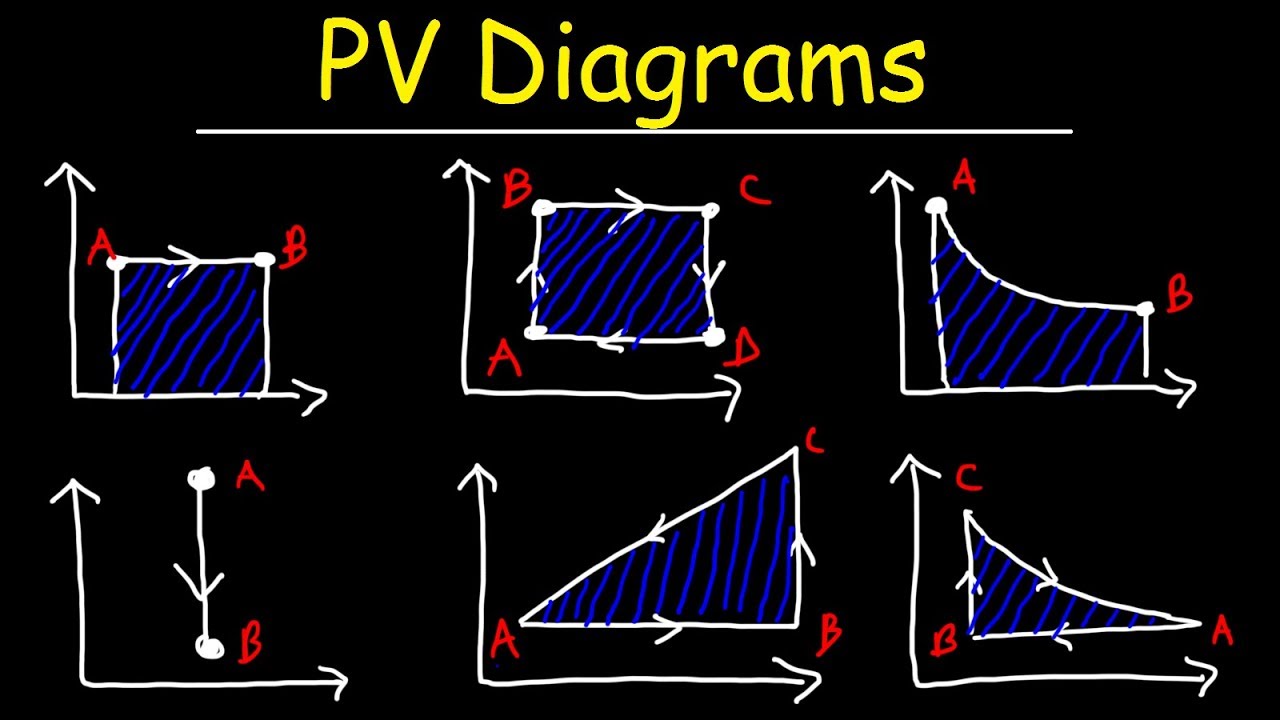 Pv Diagrams How To Calculate The Work Done By A Gas Thermodynamics
Pv Diagrams How To Calculate The Work Done By A Gas Thermodynamics
 Thermodynamic Temperature Wikipedia
Thermodynamic Temperature Wikipedia

Energy Enthalpy And The First Law Of Thermodynamics
 Solved Does This Diagram Represent An Increase Or Decreas
Solved Does This Diagram Represent An Increase Or Decreas
 Consider The Accompanying Energy Diagram Clutch Prep
Consider The Accompanying Energy Diagram Clutch Prep
 Trigonometric Model Of Opposites Two Coexisting Bifurcations
Trigonometric Model Of Opposites Two Coexisting Bifurcations
 Chapter 2a Pure Substances Phase Change Properties Updated 9 20 09
Chapter 2a Pure Substances Phase Change Properties Updated 9 20 09
 Energy Bar Charts Lol Diagrams Physics Blog
Energy Bar Charts Lol Diagrams Physics Blog
Energy Enthalpy And The First Law Of Thermodynamics
The First Law Of Thermodynamics



0 Response to "Does This Diagram Represent An Increase Or Decrease In The Internal Energy Of The System"
Post a Comment